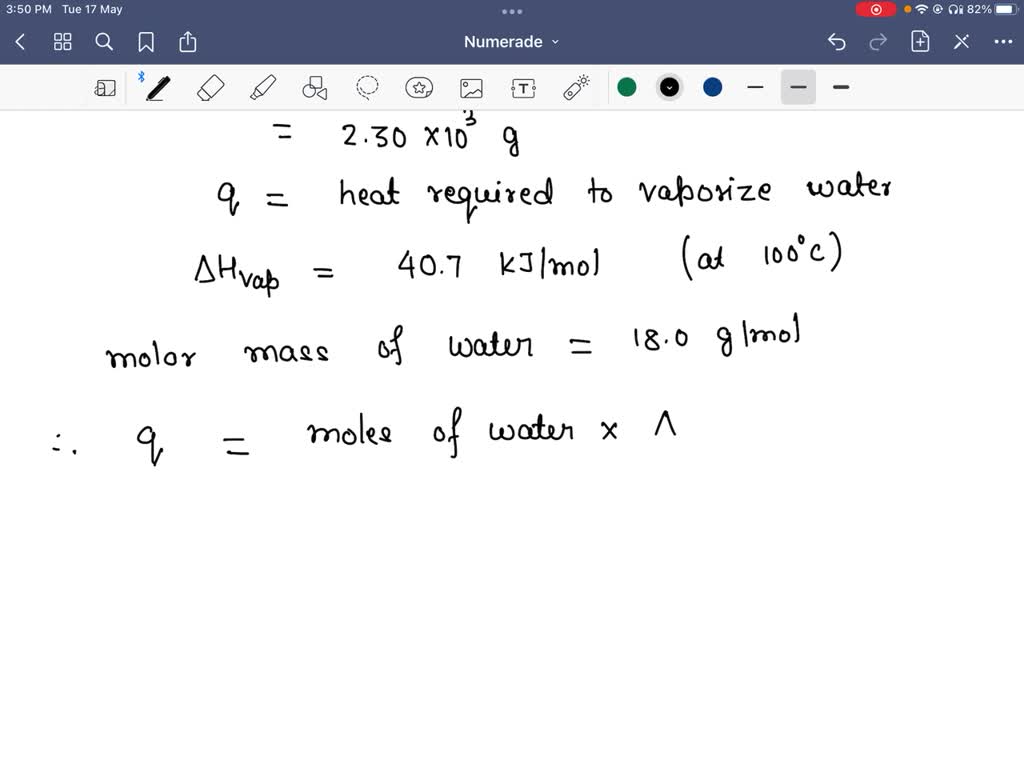
SOLVED: The mass defect for an isotope was found to be 0.410 amulatom: Calculate the binding energy in kJ/mol of atoms: (1 J = kg m2/s2 ;lamu =1.66054e-24; 1 Mole equals 6.022x

Homework 3 Solutions - Introductory Physical Chemistry | CH 331 | Assignments Physical Chemistry | Docsity
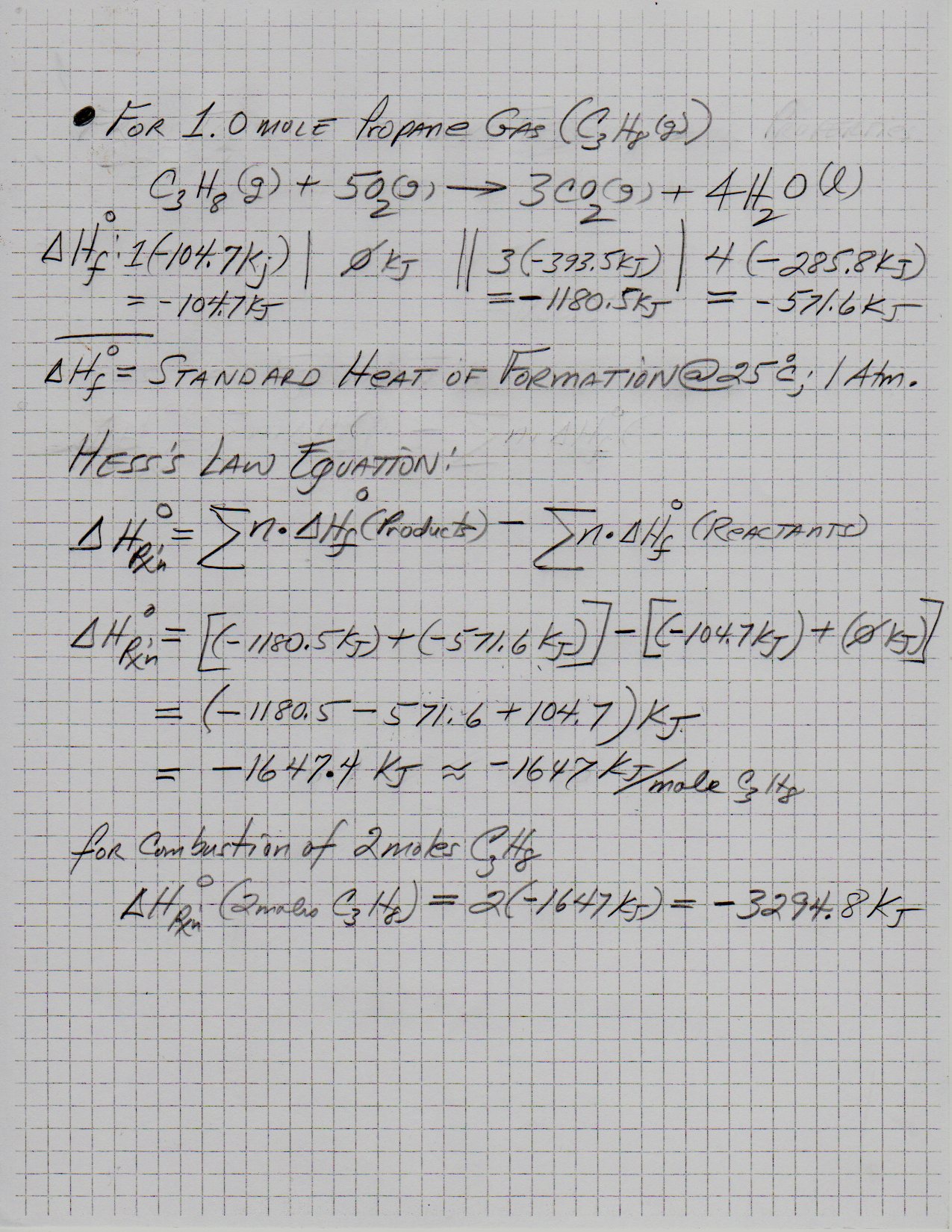
Using the chemical equation below, determine the energy released by burning 2 moles of propane, "C"_3"H"_8? a) 4438.4 kJ b) 11096 kJ c) 2219.2 kJ d) 1109.6 kJ | Socratic

The enthalpy of combustion of methane, graphite and dihydrogen at 298K are - 890.3 kJmol^-1, - 393.5 kJmol^-1, - 285.8 kJ mol^-1 respectively. Enthalpy of formation of CH4(g) will be:
The heat of combustion of C, H2 and CH4 at 298 K and 1 atm are respectively-393 kJ/mol, -286 kJ/mol, and -892 kJ/mol. How do I calculate the enthalpy of formation for