
Henry law constant for oxygen dissolved in water is` 4.34 xx 10^(4) atm` at `25^() C`. If the pa... - YouTube
-340.png)
Henry's law constant at temperature T (given Henry constant at standard temperature,enthalpy of solution)

Henry's law constant for CO2 in water is 1.67 X 10^8 Pa at 298K. Calculate the quantity of CO2 in... - YouTube

Henry law constant for the solubility of methane in benzene at 298K is 4.27 × 10^5 mm Hg then the solubility of methane in benzene at 298K under 760mm Hg is:

Henry\'s law constant for `CO_(2)` in water is `1.67xx10^(8) Pa` at `298 K`. Calculate the quant... - YouTube

Henry's law constant for the molality of methane in benzene at 298 K is 4.27 × 10^5 mm Hg . Calculate the solubility of methane in benzene at 298 K under 760 mm Hg .

SOLVED: At 25.0 *C the Henry's Law constant for argon (Ar) gas in water is 1.4 x 10` Mlatm: Calculate the mass in grams of Ar gas that can be dissolved in

The Henry's law constant for the solubility of N 2 gas in water at 298 K is 1.0 × 105 atm. The mole fraction of N 2 is 0.8. The number of





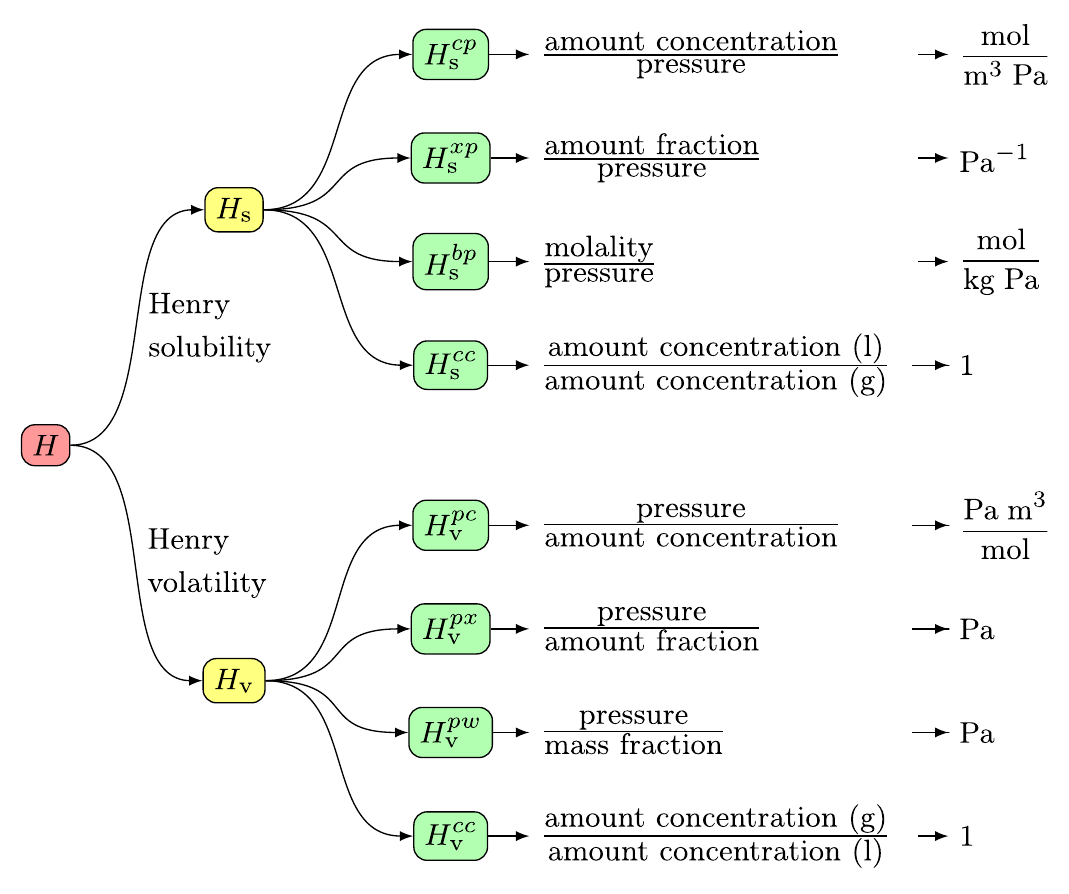

![PDF] Compilation of Henry's law constants (version 4.0) for water as solvent | Semantic Scholar PDF] Compilation of Henry's law constants (version 4.0) for water as solvent | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c61dc4a148ae9f5913ab5c8db96d120269701eb1/2-Table2-1.png)


![PDF] Compilation of Henry's law constants (version 4.0) for water as solvent | Semantic Scholar PDF] Compilation of Henry's law constants (version 4.0) for water as solvent | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c61dc4a148ae9f5913ab5c8db96d120269701eb1/6-Table5-1.png)


